|
Gold dissolves in alkaline solutions of cyanide, which are used in mining and electroplating. Gold is insoluble in nitric acid alone, which dissolves silver and base metals, a property long used to refine gold and confirm the presence of gold in metallic substances, giving rise to the term ' acid test'. Gold is resistant to most acids, though it does dissolve in aqua regia (a mixture of nitric acid and hydrochloric acid), forming a soluble tetrachloroaurate anion. Less commonly, it occurs in minerals as gold compounds, often with tellurium ( gold tellurides). 
It occurs in a solid solution series with the native element silver (as in electrum), naturally alloyed with other metals like copper and palladium, and mineral inclusions such as within pyrite. Gold often occurs in free elemental ( native state), as nuggets or grains, in rocks, veins, and alluvial deposits. It is one of the least reactive chemical elements and is solid under standard conditions. Chemically, gold is a transition metal and a group 11 element. It is a bright, slightly orange-yellow, dense, soft, malleable, and ductile metal in pure form. This makes it one of the higher–atomic-number elements that occur naturally. You can effortlessly find every single detail about the elements from this single Interactive Periodic table.Gold is a chemical element with the symbol Au (from Latin aurum 'gold') and atomic number 79. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Free Gift for you: Interactive Periodic Table Gold is also used in making medals and trophies for winners of the games.Gold is highly malleable, due to which it is used in making very thin foil that can be applied on various shapes.Gold is also used in dentistry as it is inert metal, and it is nonallergic.Hence it is used in computers, mobile phones, as well as other electronics. Gold is a very good conductor of electricity and it can conduct very small currents.In earlier days, gold was used in the manufacturing of gold coins (which was used as a currency).As gold is a precious metal, it is used in the manufacturing of jewelry and ornaments.Gold is not affected by most acids, but it dissolves in aqua regia (aqua regia is a mixture of nitric acid and hydrochloric acid).At 180 ☌, the powdered form of gold reacts with chlorine and forms AuCl 3.Gold reacts with fluorine at higher temperature and forms gold (III) fluoride. Gold does not react with oxygen even at higher temperature, but it does react with strong halogen like fluorine.But the most common oxidation states of gold are +1 and +3. The oxidation states of gold range from -1 to +5.Gold is a non reactive metal and it does not corrode or tarnish in the air.Chemical properties of GoldĬhemical properties of gold are mentioned below. 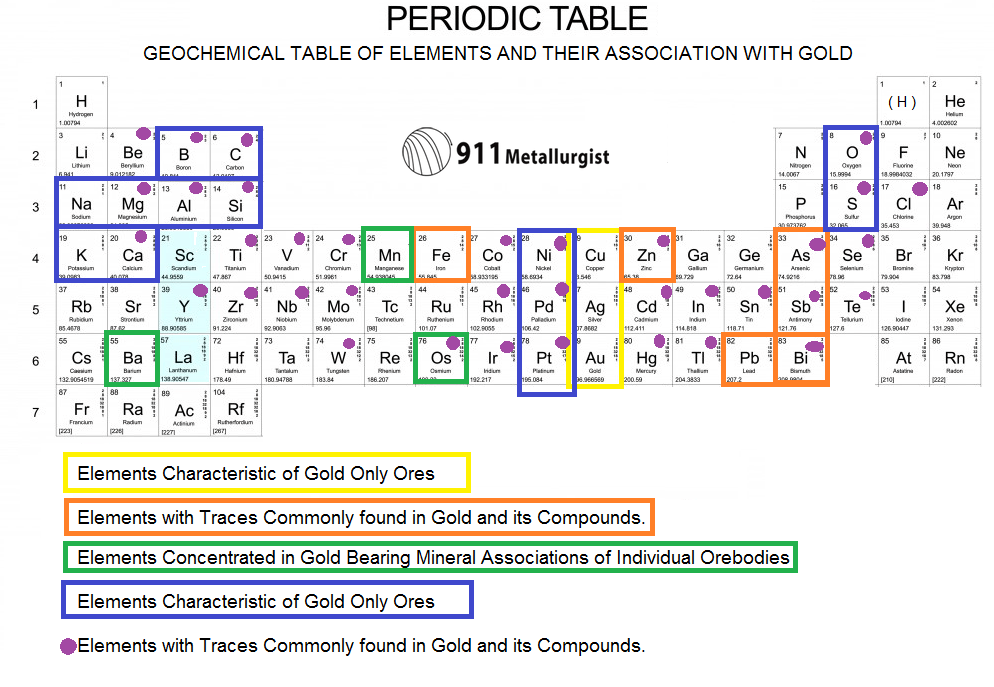
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
